  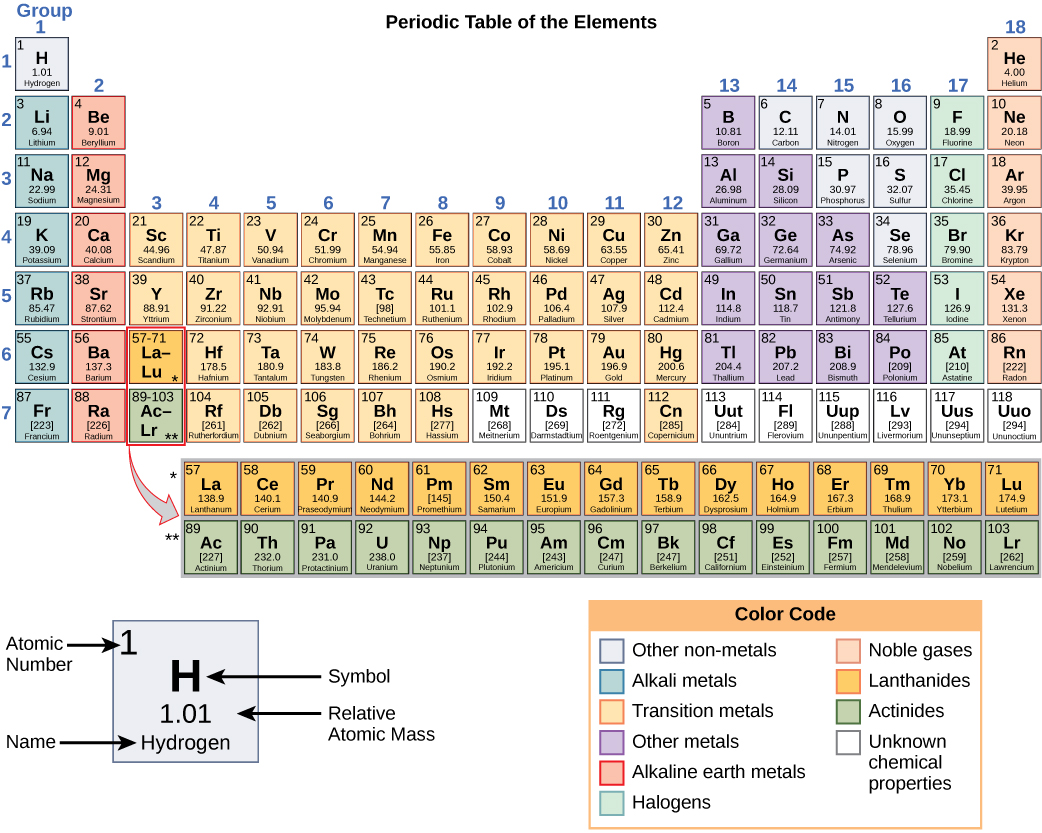
n u m b e r o f n e u t r o n s n u m b e r o f n e u t r o n sĪll atoms of oxygen-18 contain 10 neutrons. Into the mass number equation and solve for the number of neutrons:ġ 8 = 8 + 1 0 =. We can substitute the number of protons and the mass number We have already established that an atom of oxygen-18 contains eight protons. The following equation can be used to calculate the The term nucleons collectively refers to protons and neutrons. The mass number is the total number of nucleons in the nucleus of an atom, In nuclide notation, the top-left number is the mass number. Therefore, all atoms of oxygen-18 have eight protons. Of an atom, isotope, or ion of the element. The atomic number represents the number of protons in the nucleus In nuclide notation, the bottom-left number is the atomic number. How many neutrons are there in atoms of oxygen-18?.How many protons are there in atoms of oxygen-18?.The isotope oxygen-18 has the nuclide symbol 1 8 8 O. Using our understanding of atomic number and mass number, we can represent an atom of boron that contains five protons, six neutrons,Īnd five electrons with the following nuclide notation.Įxample 2: Deducing the Number of Protons and Neutrons and the Mass Number from Nuclide Notation n u m b e r o f n e u t r o n s n u m b e r o f n e u t r o n sĪn atom of calcium has 22 neutrons in its nucleus. Number given in the question into the mass number equation and solve for the number of neutrons:Ĥ 2 = 2 0 + 2 2 =. 
We can substitute the number of protons and the mass We have already established that an atom of calcium contains 20 protons. M a s s n u m b e r n u m b e r o f p r o t o n s n u m b e r o f n e u t r o n s = +. The following equation can be used to calculate the mass number: The mass number is the total number of nucleons in the nucleus of an atom, isotope, or ion. Therefore,Īn atom of calcium with an atomic number of 20 has 20 protons in its nucleus. The atomic number represents the number of protons in the nucleus of all atoms, isotopes, and ions of the element. How many neutrons are there in its nucleus? Answer Example 1: Converting from the Mass Number and the Atomic Number to the Number of NeutronsĪn atom of calcium has a mass number of 42 and an atomic number of 20. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
